Introduction
Formalin-fixed paraffin-embedded (FFPE) tissues represent an important source of archival clinical material for molecular analysis. However, nucleic acids recovered from FFPE samples are often fragmented and contain a high proportion of short DNA fragments generated during fixation and long-term storage.
The MagPure FFPE DNA Kit (D6323B) is designed for magnetic bead-based purification of genomic DNA from FFPE tissue sections. The binding chemistry enables efficient recovery of DNA fragments suitable for downstream molecular workflows while allowing selective removal of very short degraded fragments generated during FFPE processing.
This characteristic can be beneficial in workflows where excessive small DNA fragments may interfere with downstream sequencing or library preparation steps. Purified DNA can be directly used for PCR amplification, mutation analysis, or sequencing-based applications.
For laboratories requiring a column-based workflow for FFPE DNA extraction, the HiPure FFPE DNA Kit (D3126) provides a membrane purification system within the Magen FFPE product line.
For applications requiring simultaneous recovery of both DNA and RNA from the same FFPE sample, the MagPure FFPE DNA/RNA Kit (R6327) provides a magnetic bead-based co-extraction workflow.
When higher DNA purity is required for inhibitor-sensitive downstream assays, the MagPure FFPE DNA Kit (High Pure) (D6323D) offers an alternative purification chemistry optimized for removal of pigments and contaminants.
Details
Specifications
|
Features
|
Specifications
|
|
Main Functions
|
Isolation total DNA from FFPE using high bind beads
|
|
Applications
|
RT-PCR, northern blot, poly A purification, nucleic acid protection and in vitro translation, etc.
|
|
Purification technology
|
Magnetic beads technology
|
|
Process method
|
Manual or automatic
|
|
Sample type
|
Large quantities of solids
|
|
Sample amount
|
Appropriate
|
|
Elution volume
|
≥50μl
|
|
Time per run
|
30 - 120 minutes
|
Extraction Principle
Samples are first deparaffinized and digested with Proteinase K to release nucleic acids from crosslinked tissue matrices. Heat treatment partially reverses formaldehyde-induced crosslinking, improving DNA accessibility.
Following lysis, DNA molecules bind to magnetic particles under chaotropic salt conditions. By adjusting the amount of binding buffer during the adsorption step, fragmented DNA molecules within the range of approximately 100–300 bp can be selectively removed from the lysate. This selective binding strategy enriches longer DNA fragments that are more suitable for downstream sequencing analysis.
After washing steps to remove contaminants, purified DNA is eluted in a low-salt buffer suitable for molecular biology workflows.
Kit Contents
|
Contents
|
D632301B
|
D632302B
|
|
Purification Times
|
48 Preps
|
96 Preps
|
|
MagBind Particles
|
1.1 ml
|
2 x 1.1 ml
|
|
RNase A
|
10 mg
|
20 mg
|
Proteinase K
|
24 mg
|
48 mg
|
Protease Dissolve Buffer
|
3 ml
|
6 ml
|
|
Buffer DPS
|
60 ml
|
100 ml
|
|
Buffer ATL
|
15 ml
|
30 ml
|
|
Buffer AL
|
15 ml
|
30 ml
|
|
Buffer BD*
|
6 ml
|
15 ml
|
Buffer BXW1*
|
13 ml
|
44 ml
|
|
Elution Buffer
|
15 ml
|
30 ml
|
Storage and Stability
RNase A, Proteinase K and MagBind Particles should be stored at 2-8°C upon arrival. However, short-term storage (up to 12 weeks) at room temperature (15-25°C) does not affect their performance. The remaining kit components can be stored at room temperature (15-25°C) and are stable for at least 18 months under these conditions.
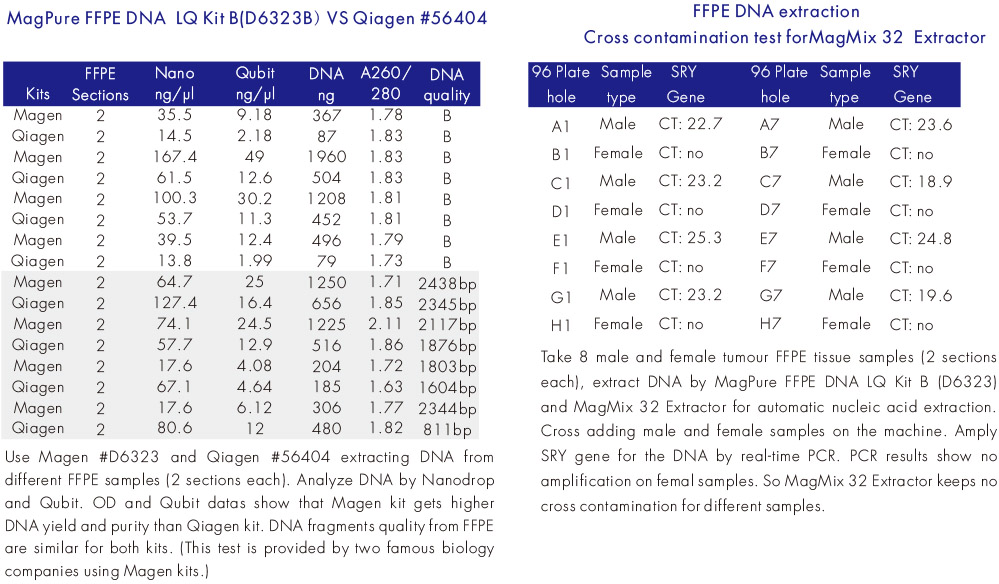
Experiment Data
Purchase Guide
For guidance on selecting the most appropriate FFPE nucleic acid extraction system based on target analyte, workflow format and downstream application requirements:
👉 FFPE Nucleic Acid Extraction Purchase Guide
For a broader technical overview of FFPE DNA, RNA and DNA/RNA co-extraction workflow routes, processing logic and application-oriented route design:
👉 FFPE Nucleic Acid Extraction Workflows Explained
For detailed workflow structure, estimated processing time and route-specific handling logic across representative FFPE workflows: